 Protonix (pantoprazole) is a proton-pump inhibitor (PPI) used in the treatment of gastroesophageal reflux disease (GERD) and Zollinger-Ellison Syndrome. There have been reports of patients using this drug who have tested “positive” in drugs of abuse screens for marijuana (tetrahydrocannabinol or THC). These reports appear to be limited only to screening tests for cannabinoids and only with some – not all – of the immunoassays (screening tests) for cannabinoids. The manufacturer of Protonix, Wyeth Pharmaceuticals, in their package insert for the drug is available online and states the following:
Protonix (pantoprazole) is a proton-pump inhibitor (PPI) used in the treatment of gastroesophageal reflux disease (GERD) and Zollinger-Ellison Syndrome. There have been reports of patients using this drug who have tested “positive” in drugs of abuse screens for marijuana (tetrahydrocannabinol or THC). These reports appear to be limited only to screening tests for cannabinoids and only with some – not all – of the immunoassays (screening tests) for cannabinoids. The manufacturer of Protonix, Wyeth Pharmaceuticals, in their package insert for the drug is available online and states the following:
7.4 False Positive Urine Tests for THC
There have been reports of false positive urine screening tests for tetrahydrocannabinol (THC) in patients receiving proton pump inhibitors. An alternative confirmatory method should be considered to verify positive results.
Quest Diagnostics is not aware of any reports of false positive screens for marijuana with the technology utilized in our screening tests for cannabinoids. Moreover, the chain of custody workplace tests for the drugs of abuse, including marijuana, all utilize gas chromatography/mass spectrometry (GC/MS) or equivalent technology as the confirmatory method to definitively identify and quantify the drug or drug metabolite present in the donor’s specimen. Consequently, the use of Protonix should not result in a confirmed positive drug test for marijuana or its metabolite.
For more information about drug testing, visit our website or contact us online.

 Your Privacy Choices
|
Privacy Notices
|
Terms
|
Language Assistance / Non-Discrimination Notice | Asistencia de Idiomas / Aviso de no Discriminación | 語言協助 / 不䈚視通知
Your Privacy Choices
|
Privacy Notices
|
Terms
|
Language Assistance / Non-Discrimination Notice | Asistencia de Idiomas / Aviso de no Discriminación | 語言協助 / 不䈚視通知





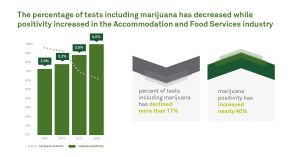


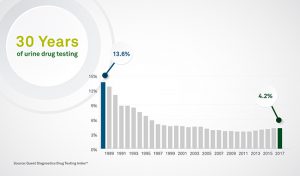

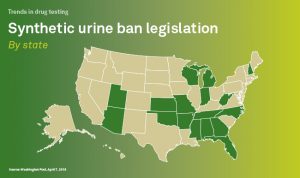







7.4 False Positive Urine Tests for THC
There have been reports of false positive urine screening tests for tetrahydrocannabinol (THC) in patients receiving proton pump inhibitors. An alternative confirmatory method should be considered to verify positive results.
Quest Diagnostics is not aware of any reports of false positive screens for marijuana with the technology utilized in our screening tests for cannabinoids. Moreover, the chain of custody workplace tests for the drugs of abuse, including marijuana, all utilize gas chromatography/mass spectrometry (GC/MS) or equivalent technology as the confirmatory method to definitively identify and quantify the drug or drug metabolite present in the donor’s specimen. Consequently, the use of Protonix should not result in a confirmed positive drug test for marijuana or its metabolite.
For more information about drug testing, visit our website or contact us online.